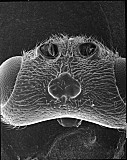
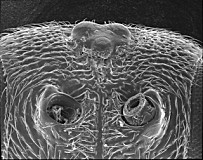
Fopius arisanus (Sonan, 1932)
Prior to the description of Fopius, the species now placed in this genus were most frequently included either in Opius or Biosteres. This particular species has also been placed, occasionally, in Diachasma. Thus, the names Opius oophilus, Biosteres oophilus, and Diachasma oophilus all refer to Fopius arisanus.
Fullaway described Opius oophilus on the basis of specimens collected in Hawaii following the introduction of this species against Oriental fruit fly in the late 1940s, but oophilus was later recognized as a synonym of arisanus, originally described from Taiwan. The actual country of origin for the material introduced to Hawaii is uncertain (though believed to be Malaysia). Fopius arisanus has subsequently become established in several other countries, including Australia, Costa Rica, Fiji, and Mauritius (Wharton et al. 1981, Wharton and Gilstrap 1983) as a result of purposeful redistribution from Hawaii targeting a variety of pest tephritids (Wharton 1989). For a more recent summary, see Rousse et al. (2005).
Fopius arisanus has achieved a competitive advantage over other introduced parasitoid species in Hawaii. Wang and Messing (2003) have shown several mechanisms by which F. arisanus optimizes its parasitization capabilities including adjusting foraging time according to patch quality cues (presence of plant injury odors, kariomones, and previous experience in patch), utilizing a fixed set of host location behaviors (antennating, probing, detecting, and ovipositing), and discriminating nonparasitized from parasitized hosts for oviposition.
Similar to results from previous experiments with Diachasmimorpha tryoni (Wang and Messing 2003), Diachasmimorpha krausii, and Psyttalia concolor (Wang and Messing 2002), Wang et al. (2003) found that F. arisanus was the superior competitor when matched with Diachasmimorpha longicaudata. As an egg parasite, F. arisanus arrives on the host (Ceratitis capitata and Bactrocera dorsalis in these experiments) prior to D. longicaudata, a larval parasite, and suppresses development of D. longicaudata, primarily at the egg stage. The authors also report that in experiments with the 4 larval parasitoids none were able to directly destroy F. arisanus larvae during parasitism. Although a superior competitor among many fruit fly parasitoids, F. arisanus does not flourish in all environments including higher elevations and lower temperatures, and is not adapted to certain tephritid hosts including Bactrocera cucurbitae. These recent studies verify and expand on the earlier studies of interspecific competition by van den Bosch and Haramoto (1951), van den Bosch and Haramoto (1953) and Bess et al. (1961). For a detailed review of the biology of this species, see Rousse et al. (2005).
Other experimental studies provide evidence that F. arisanus is able to successfully parasitize and complete development in olive fly, Bactrocera oleae, as well as cause incidental fly egg mortality through ovipositor insertion (Calvitti et al. 2002). Similar results were obtained on Anastrepha suspensa after earlier failures (Lawrence et al. 2000) and on other species of Anastrepha (Zenil et al. 2004). Snowball and Lukins (1964) reared arisanus from several species of Bactrocera when it was introduced to Australia for biological control of Queensland fruit fly. Chinajariyawong et al. (2000) provide a long list of native hosts from Thailand and Malaysia.
The potential impact of F. arisanus and 2 other tephritid egg parasitoids (F. caudatus and F. ceratitivorus) on a nontarget flowerhead-feeding tephritid, Trupanea dubautiae, was evaluated by Wang et al. (2004). Trupanea dubautiae is critical for pollination of plants in Hawaii. In laboratory experiments, no evidence of parasitism of T. dubautiae by any of the 3 parasitoids was observed.
Efficacy in combination with SIT: Vargas et al. (1994) and Vargas (1996) and with bait sprays: Vargas et al. (2001).
Suggestions for augmentative biological control: Harris et al. (2000).
Potential for use against olive fly in California: Sime et al. (2008).
Introduced to Society Islands (Tahiti, Moorea, Raiatea, Tahaa, and Huahine) from Hawaii from 2002-2004, resulting in the most successful example of classical biological control of tephritid fruit flies in the Pacific area outside of Hawaii (Vargas et al. 2007).
A listing of F. arisanus introductions in the New World for biological control is provided below summarized from Ovruski, Aluja, Sivinski, and Wharton 2000:
Mexico—introduced in 1954 against Anastrepha ludens; Jimenez-Jimenez 1956.
Costa Rica—introduced in 1955 against Ceratitis capitata; specimens recovered, established (Holler et al. 1996, Wharton et al. 1981, Wharton et al. 1998).
Argentina—introduced in 1961 against C. capitata; Ovruski 1995.
Florida, USA—introduced 1974-75 against A. suspensa; Baranowski et al. 1993.
See also Rousse et al. 2005.